Biogenic calcium carbonate solubilities through lab and field saturometry
|
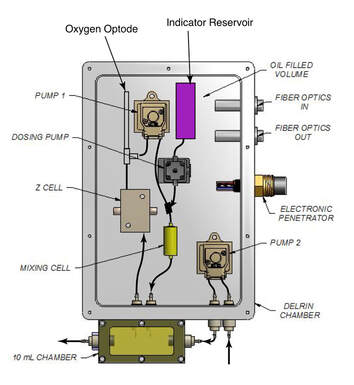
We do not fully understand the thermodynamic controls on calcite solution in the ocean. For example, the pressure dependence of calcite solubility has never been successfully reproduced in the field. I am building a saturometer, which will test the reactivity and solubility of biogenic calcites and aragonites in the lab and the field across a range of heretofore underexplored thermodynamic variables. This device is being redesigned with a spectrophotometric pH sensor. This research is the focus of a funded NSF Chemical Oceanography proposal with co-P.I. Matt Long at WHOI.
|
Coccoliths and foraminifera and their contribution to the marine CaCO3 cycle
Coccolithophores and foraminifera occupy distinct ecological niches, and therefore contribute in distinct ways to the production and export of calcium carbonate. The large inventory of calcium carbonate on the seafloor is made up of coccoliths and foraminifera; however, the relative contribution of coccoliths and foraminifera to sedimentary calcium carbonate is not constrained. This project involves investigating changes in calcium carbonate production and dissolution using size-fractionated measurements of calcium carbonate content and d13C in sediment cores over the last million years.
Impacts and biological feedbacks associated with ocean alkalinization
|
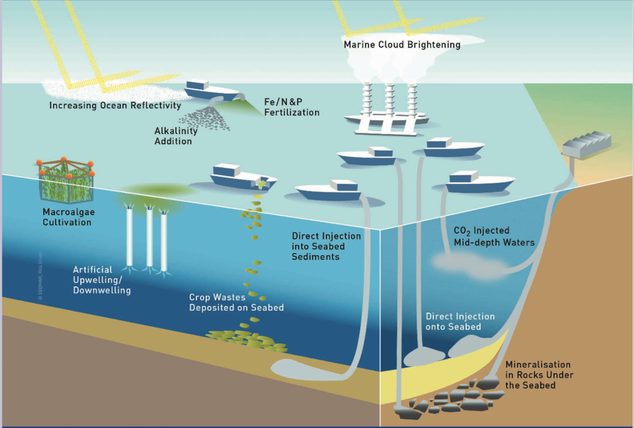
Ocean alkalinization is an approach to mitigating anthropogenic CO2 emissions that involves increasing the already substantial buffering capacity of the world oceans. Despite being one of the few approaches that can sequester all anthropogenic emissions, there is little research being done on the impacts and biological feedbacks of ocean alkalinization. B.B. Cael at Southampton and I are working on assessing these impacts through culture and incubation experiments, utilizing 13C-labeled bicarbonate to track more carefully how and where carbon is moving through the ecosystem. Image is from the 2019 GESAMP working group on ocean alkalinization.
|
Calcite dissolution kinetics using isotopically labeled carbon, calcium, and magnesium
|
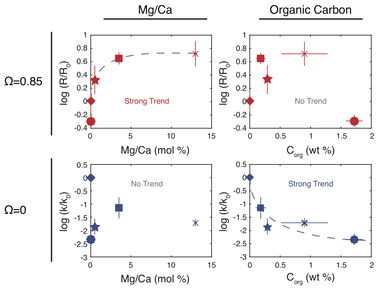
We have developed a new method to measure the dissolution kinetics of calcite in seawater. Instead of tracking a bulk chemistry change, we dissolve Carbon-13 labeled calcium carbonate in natural seawater, and mass transfer between solid and solution is traced by measuring the increase in the 13/12 carbon ratio of seawater over time. This work has revealed mechanistic insights into the nature of calcite dissolution in seawater, and the controls on how biogenic calcites dissolve relative to inorganic calcite. I am interested in extending this technique to Mg and Ca isotopes in order to investigate the reaction kinetics of high-Mg calcites; in particular fish carbonates. This work is in collaboration with Michael Salter at the University of Exeter.
|